Teva announces FDA approval of Austedo tablets for chorea associated with huntington's disease - Pharma Advancement
Austedo (deutetrabenazine) for the Treatment of Chorea Associated with Huntington's Disease - Clinical Trials Arena

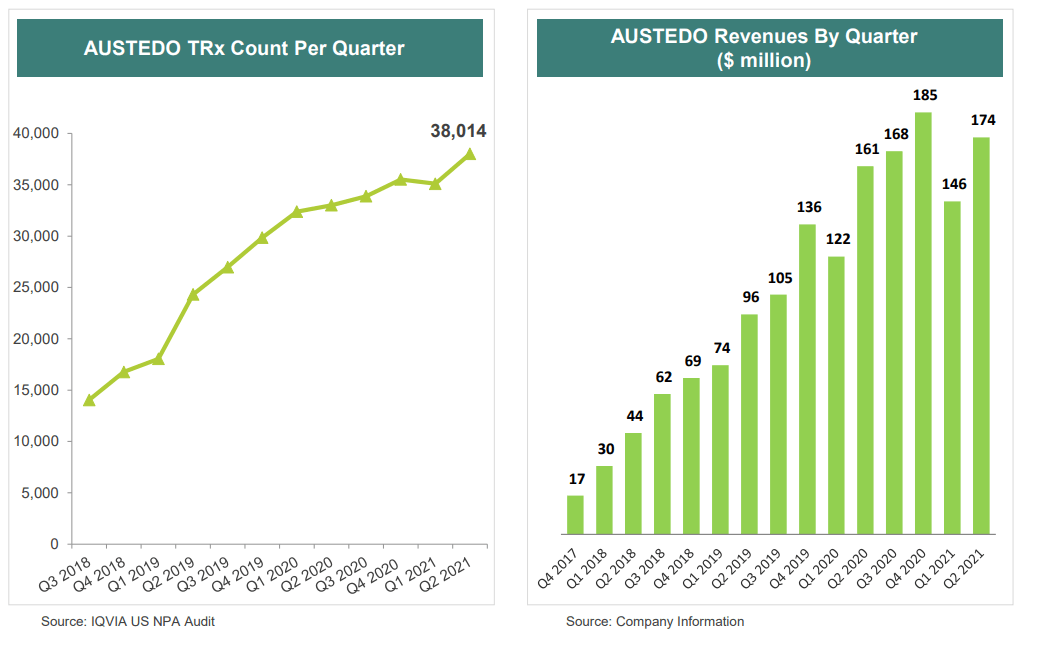
Punit on Twitter: "#Austedo stellar Q2 sales reported by Teva on 5th Aug'20 Austedo net sales in FY19 stood $412M & continues to grow in H1FY20👇 Q2-17- $1 M Q2-18- $44 M

Teva Announces FDA Approval of AUSTEDO™ (deutetrabenazine) Tablets for the Treatment of Chorea Associated with Huntington's Disease — Hereditary Neurological Disease Centre

Teva's Austedo (deutetrabenazine) Receives the NMPA's Approval to Treat Chorea Associated with Huntington's Disease and Tardive Dyskinesia in Adults